
In 1999 lovastatin, a blockbuster cholesterol drug with annual peak sales of more than $1 billion in the U.S., became commercially available in Egypt — twelve years after it was first approved for sale by the U.S. Food and Drug Administration. Our research shows that this is not exceptional — long launch lags are common and 45 per cent of all new drugs are only launched in ten or fewer countries within a decade. Since delayed launch means foregone health benefits, it is important to understand how public policy affects the diffusion of new drug innovations. Our research demonstrates that the patent and price regulation policies governments adopt have a powerful impact on the speed at which new drugs become available in different countries, and this is true both for countries at high and low levels of economic development.
Promoting affordable access to new drugs is a central objective of government policy. This poses two challenges: providing adequate incentives for the development of new drugs, and ensuring affordable prices of drugs once developed. Governments use two main instruments to achieve these goals: patents and price regulation. Economics emphasizes a basic trade-off between the welfare gains from stronger innovation incentives provided by patents and the welfare loss created by the resulting higher prices. Reflecting this concern, most research on patents and `access’ to drugs has focused on how the 1994 TRIPS Agreement, which mandated global harmonization of pharmaceutical patent rights, affected prices in emerging markets.
In the debates over TRIPS and, more recently, the Trans-Pacific Partnership trade agreement, developing countries and public health advocacy groups argued that harmonization of patent policy is both unnecessary and harmful when viewed from the perspective of this trade-off. For low-income countries with limited private health insurance and poorly funded public health systems, the welfare loss involves not just the deadweight loss from higher prices, but also the worrying prospect that large segments of the population may have no affordable access to new drug therapies. This has led economists to recommend alternative ways for governments to provide innovation incentives while maintaining low prices in developing countries, especially for vaccines. Moreover, the increase in innovation incentives from having patent rights in low-income countries is likely to be small for many kinds of drugs because these countries do not account for a large part of the global market.
This debate, however, misses a critical element: the impact patent rights and other policies have on the diffusion of new drugs. The public health benefits of new drugs depend, first, on how quickly (if at all) drugs are launched in different countries and, second, on how widely they are adopted within a country, once launched. The decision to launch in any given country will be sensitive to drug manufacturers’ assessment of anticipated profits relative to country-specific costs. These include costs of clinical trials to secure regulatory approval and commercial costs relating to product launch, such as establishing distribution capacity, educating prescribers, and getting reimbursement from private or public insurers. These costs must be incurred in every country in which a drug is launched: outside tightly integrated trading blocs such as the European Union, there are few international protocols that recognize regulatory approval of drugs across borders. Moreover, the bulk of these entry costs would apply whether the first entrant in a country is the original innovator, its licensee or a generic imitator.
If the sunk investments required to enter and penetrate new markets are significant, the diffusion of new technologies will also be influenced by policies that affect profitability of suppliers in different markets, including patent rights. Of course, the potential role of patent rights in promoting global diffusion of innovation is not limited to pharmaceuticals, but they are a good case study both because of their economic importance and the significant, country-specific costs of launching new drugs.
In this research we study how patent and price control policies, as well as economic and demographic factors, affect the speed and scope of diffusion of new pharmaceutical products across countries. The empirical analysis is based on a large data set that covers launches of 642 new drugs (chemical molecules) in up to 76 countries during the period 1983-2002, together with information on the patent and price control regimes in these countries. The data cover countries that span all levels of economic development and exhibit a wide variety of patent regimes. In the analysis we distinguish between process patents and product patents on new chemical molecules. Process patents are considered relatively weak, as they do not prevent competitive entry by entrants with superior manufacturing processes. Some countries (such as India) purposefully adopted a `process only’ patent regime for drugs in order to foster domestic competitive entry. Product patents are typically considered stronger rights, blocking entry by competitive (or generic) products and allowing more effective appropriation of profit.
Our research demonstrates four main findings. First, the global diffusion of new drugs is slow. Many new drugs become available in countries only after long lags (often more than 10 years) from the date at which they were first launched commercially, and many drugs are never launched outside a handful of wealthier countries.
Second, patent policies governments adopt strongly affect how quickly new drug therapies are launched in their countries. Longer, and stronger, patent protection powerfully accelerates diffusion. For example, controlling for economic and demographic factors, moving from a regime of no product patents to a long product patent term reduces launch lags by about 55 percent. Process patents also promote faster launch, but the impact is not as large as for product patents. Short product patents have no effect. Importantly, we show that the impact of policy regimes holds equally for low and middle-income countries as for high income countries.
Third, countries that adopt strong pharmaceutical price controls experience significantly longer launch lags for new drugs. We estimate that introducing price controls increases launch lags by between 25-80 per cent. Finally, we find that local market size — as captured by population, per capita income, health expenditures, and demographic factors — has a big impact on the speed of drug launches.
Of course, the same policies that promote faster launch — stronger patent rights and the absence of price regulation — are also those that raise prices. This highlights the basic trade-off countries face between making new drug therapies available and making them affordable. Finding ways to mitigate the adverse effects of this trade-off remains a major challenge. One possible approach would be to introduce multilateral recognition of drug trials and regulatory approval, lowering launch costs and speeding up global drug diffusion. Finally, our paper highlights the broader point, not limited to pharmaceuticals, that patent rights can have an important impact on the diffusion of new innovations as well as on the rate at which new innovations are created.
Table 1. Global diffusion of new drug launches
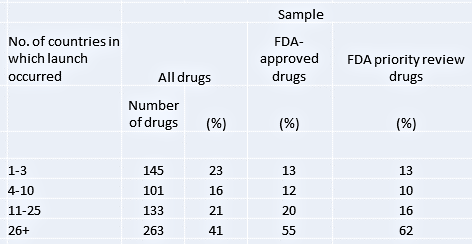
Table 1 presents information showing how many countries drugs were launched in (the geographic scope of diffusion). The striking fact is that diffusion of new drugs around the world is remarkably limited. For the entire sample of new drugs, 39 per cent were launched in ten or fewer countries during the sample period, and only 41 per cent were launched in more than 25 countries. The average number of countries experiencing launch is 22.4 out of a possible 76. Even among the wealthier countries with the most developed health care systems, not all drugs became available during the sample period: e.g., only about 60 per cent were launched in the U.S., Germany or the U.K. This limited geographic diffusion suggests a potentially substantial welfare loss. Even among the set of ‘high-quality’ drugs (those that were priority-reviewed by the U.S. Food and Drug Administration, FDA), 13 percent of new drugs were launched in no more than three countries within the sample period.
The problem is just as bad when we turn to how long it takes drugs to be launched. For all drugs and countries together, we find that after 10 years only 41 per cent of drug-country opportunities for a launch were taken up (not shown here). Even after 20 years or more, less than 50 per cent of possible launches had taken place, and as practical matter, many of these drugs may never be launched in large numbers of countries. While not all delays necessarily represent welfare losses (some drugs may have become obsolete, may have no value in contexts where important complementary technologies or resources for health care are not available, or may only be useful for treating diseases with low incidence in a country), this evidence of limited diffusion is nonetheless disappointing from a welfare perspective. Even in the sub-sample of FDA-approved drugs, only 54 per cent were launched in the average country within 10 years. Diffusion of non-FDA-approved drugs was much slower and less extensive, with 19 per cent of drug-country launch opportunities filled within 10 years.
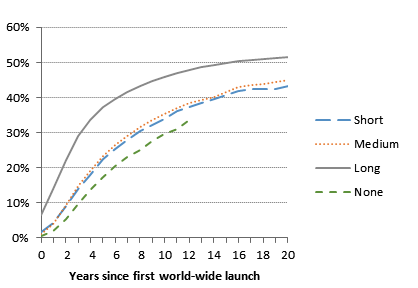
Figure 1. Fraction of drugs launched by patent regime
Figure 2. Fraction of drugs launched by price controls
Second, the speed and extent of diffusion is strongly associated with a country’s patent and price regulation regimes. Figure 1 shows results broken out by a summary measure of each country’s patent regime – None, Short (less than 10 years), Medium (11-16 years) and Long (17 years or longer). With no patents, the estimated time for 25 percent of drug-country launch opportunities to be filled is eight years, falling to less than 2.6 years with long-duration patents. Figure 2 shows that in countries with no or weak price regulation, 25 per cent of launch opportunities are filled within three years, compared to five years where price regulation is strong.
These simple comparisons tell the story. Of course, our research uses modern econometric methods to confirm these basic findings when we control for a wide range of economic, demographic and institutional factors that may also influence diffusion speed.
This research broadens the intellectual perspective and policy discussion on patents significantly, because the focus has always been limited to the how patents affect the incentives for innovation on the one side, and prices on the other. Our research shows that patent rights also affect the availability in different markets. Importantly, this impact would be expected to hold for any products where there are significant costs of launching in different markets. This identifies an important new element that needs to be taken into account in international trade agreements that cover intellectual property.
♣♣♣
- This blog post is based on the author’s paper Patents and the Global Diffusion of New Drugs, co-authored with Iain M. Cockburn and Jean O. Lanjouw, American Economic Review (2016)
- The post gives the views of its author, not the position of LSE Business Review or the London School of Economics.
- Featured image credit:
- pills flying: https://pixabay.com/en/pill-capsule-flying-many-horde-1884777/
- world map: https://pixabay.com/en/world-map-pill-earth-healthcare-1185076/
- Before commenting, please read our Comment Policy
 Mark Schankerman is professor of economics at LSE, a research fellow at the Centre for Economic Policy Research (CEPR), and Sackler Fellow, Tel Aviv University. His research interests are in intellectual property rights, innovation, industrial economics and emerging economies. He wrote the book The Comingled Code: Open Source and Economic Development, with Josh Lerner, (MIT Press, October 2010 and paperback in 2013), reviewed by The Economist in 2011.
Mark Schankerman is professor of economics at LSE, a research fellow at the Centre for Economic Policy Research (CEPR), and Sackler Fellow, Tel Aviv University. His research interests are in intellectual property rights, innovation, industrial economics and emerging economies. He wrote the book The Comingled Code: Open Source and Economic Development, with Josh Lerner, (MIT Press, October 2010 and paperback in 2013), reviewed by The Economist in 2011.







would have been helpful to include examples